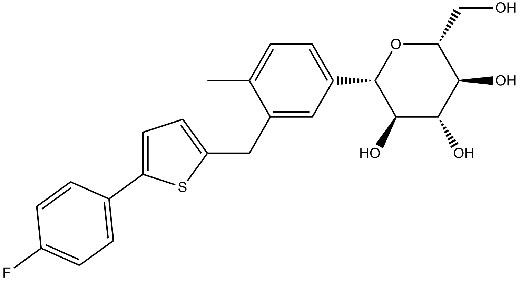
Canagliflozin
API’s Name:Canagliflozin
CAS No.:842133-18-0 & 928672-86-0
Indication:Type 2 diabetes
Innovator:
Specification:In-House
US DMF:√
EU DMF:
CEP:
Only for R&D purpose
Product Detail
Intermediates
Code | CAS | Specification |
CLF | 928672-86-0 | In-House |
CLF-2 | 1030825-20-7 | In-House |
CLFM3 | 32384-65-9 | In-House |
Background
Canagliflozin is a novel, potent, and highly selective sodium glucose co-transporter (SGLT) 2 inhibitor [1]. It has been proved that Canagliflozin can increase urine glucose excretion by reducing the renal glucose threshold and by decreasing the filtered glucose re-absorption [2].
Canagliflozin has been shown to inhibit the Na+-mediated 14C-AMG intakes in CHO-hSGLT2, CHO-rat SGLT2 and CHO-mouse SGLT2 with IC50 values of 4.4, 3.7 and 2.0 nM, respectively [1].
Canagliflozin has been reported to reduce the blood glucose (BG) levels dose-dependently in both db/db Mice and Zucker diabetic fatty (ZDF) Rats. Additionally, canagliflozin has proved to decrease the respiratory exchange ratio, and body weight in DIO mice and ZDF rats [1].
Canagliflozin can be taken orally [1].
References:
[1] Liang Y1, Arakawa K, Ueta K, Matsushita Y, Kuriyama C Martin T, Du F, Liu Y, Xu J, Conway B, Conway J, Polidori D, Ways K, Demarest K. Effect of canagliflozin on renal threshold for glucose, glycemia, and body weight in normal and diabetic animal models. PLoS One. 2012;7(2):e30555
[2] Sarnoski-Brocavich S, Hilas O. Canagliflozin (Invokana), a Novel Oral Agent For Type-2 Diabetes. P T. 2013 Nov;38(11):656-66
Product Citation
Bahia Abbas Moussa, Marianne Alphonse Mahrouse, et al. "Different resolution techniques for management of overlapped spectra: Application for the determination of novel co-formulated hypoglycemic drugs in their combined pharmaceutical dosage form." Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy Available online 20 June 2018.
Chemical structure